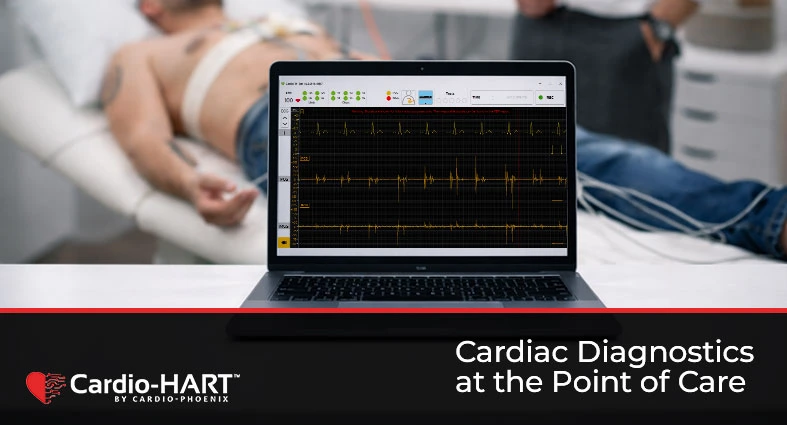
Cardio-HART: The AI-Powered Evolution of the ECG for Primary Care
Cardio-HART heart failure detection in primary care addresses one of the biggest limitations of standard ECG: the inability to reliably identify many structural and functional heart diseases during early patient presentation. Why Primary Care Needs More Than a Standard ECG Every day in Primary Care, clinicians see patients with symptoms that may suggest underlying cardiovascular […]
Cardio-HART: The AI-Powered Evolution of the ECG for Primary Care Read More »